 device malfunction, lead/electrode break or migrationīiological, e.g. Paddle: placed surgically via laminectomy or laminotomyĭevice-related, e.g. Parameters chosen for optimal pain control include burst, high density or high dose, and high frequency electrical stimuli. 
Two types of electrodes can be implanted 2:Ĭylindrical: placed via percutaneous technique under fluoroscopy Standard techniques for spinal cord stimulation for pain involve fixed-current open-loop dorsal column electrical stimuli. Abbotts Invisible Trial System uses a discreet external device to deliver the same therapy you would receive with an implanted neurostimulator. ProcedureĪn internal pulse generator is implanted in the subcutaneous tissues of the flank, paraspinal, abdominal or upper gluteal regions with leads extending into the spinal canal with epidural electrodes placed adjacent to the dorsal columns. If your pain doctor says youre a candidate for one of Abbotts neurostimulation therapies, the first step is a trial with a temporary system. 
Patients may feel a tingling sensation in the area of electrodes placement when the stimulator is on. Although it varies between individuals, generally it reduces pain sensation by ~60% 1. The main difference between the dorsal root ganglion (DRG) stimulator and the spinal cord stimulator (SCS) is the target of their respective lead wires and placement of electrodes. The underlying pathophysiology is not entirely understood, however, increase in levels of GABA and serotonin stimulated by an electrical pulse in the area of dorsal horns is one of the potential mechanisms 1. It uses low voltage electrical current delivered by electrodes on the surface of the cord to prevent pain signal from reaching the brain. SCS is a consideration for people who have a pain. failed back syndrome, brachial plexopathy, complex regional pain syndrome). A spinal cord stimulator ( SCS) or dorsal column stimulator ( DCS) is a type of implantable neuromodulation device (sometimes called a 'pain pacemaker') that is used to send electrical signals to select areas of the spinal cord (dorsal columns) for the treatment of certain pain conditions. Refer to for product manuals for complete indications, contraindications, warnings, precautions and potential adverse events.Spinal cord stimulators, also known as dorsal column stimulators, are surgically placed devices to aid with symptom relief in individuals with chronic neurological pain (e.g. Adverse events may result in fluctuations in blood glucose in patients with diabetes. May include: undesirable change in stimulation (uncomfortable, jolting or shocking) hematoma, epidural hemorrhage, paralysis, seroma, infection, erosion, device malfunction or migration, pain at implant site, loss of pain relief, and other surgical risks. Recharging a rechargeable neurostimulator may result in skin irritation or redness near the implant site. Avoid activities that put stress on the implanted neurostimulation system components. 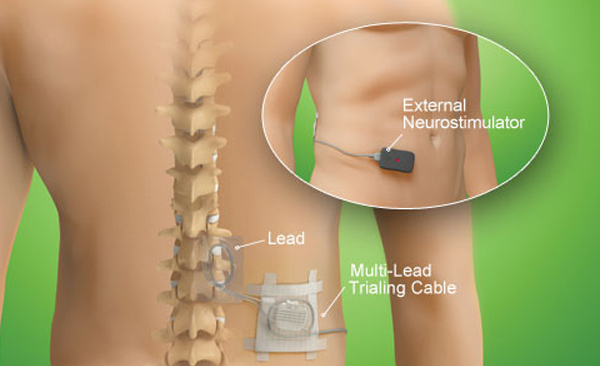
Safety and effectiveness has not been established for pediatric use, pregnancy, unborn fetus, or delivery. Neuromodulation has been used in medicine for over 50 years, with the first dorsal column stimulator implant performed in 1967 by Dr. A preoperative assessment is advised for some patients with diabetes to confirm they are appropriate candidates for surgery. Patients with diabetes may have more frequent and severe complications with surgery. Magnetic resonance imaging (MRI) revealed an edematous area in the left spinal cord between T11-T12. An implanted cardiac device (e.g., pacemaker, defibrillator) may damage a neurostimulator, and electrical pulses from the neurostimulator may cause inappropriate response of the cardiac device. Background Preclinical models of spinal cord stimulation (SCS) are lacking objective measurements to inform translationally applicable SCS parameters. She also complained of pain, weakness and numbness in both legs (left more than right). 
Sources of electromagnetic interference (e.g., defibrillation, electrocautery, MRI, RF ablation, and therapeutic ultrasound) can interact with the system, resulting in unexpected changes in stimulation, serious patient injury or death. Spinal cord stimulation (SCS) is indicated as an aid in the management of chronic, intractable pain of the trunk and/or limbs-including unilateral or bilateral pain.ĭiathermy - Energy from diathermy can be transferred through the implanted system and cause tissue damage resulting in severe injury or death. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
